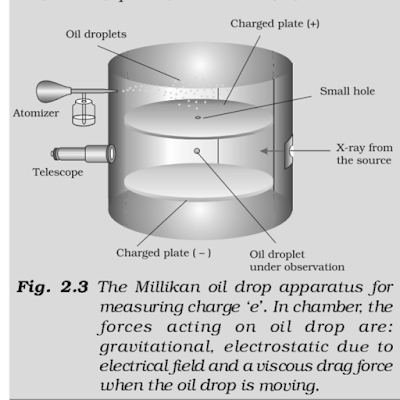
Millikan’s Oil Drop Experiment
Millikan’s Oil Drop Experiment
In this
method, oil droplets in the form of mist, produced by the atomizer, were
allowed to enter through a tiny hole in the upper plate of electrical condenser.
The downward motion of these droplets was viewed through the telescope,
equipped with a micrometer eye piece. By measuring the rate of fall of these
droplets, Millikan was able to measure the mass of oil droplets. The air inside
the chamber was ionized by passing a beam of X-ray through it. The electrical
charge on these oil droplets was acquired by collisions with gaseous ions. The fall
of these charged oil droplets can be retarded, accelerated or made stationary
depending upon the charge on the droplets and the polarity and strength of the
voltage applied to the plate. By carefully measuring the effects of electrical
field strength on the motion of oil droplets, Millikan concluded that the
magnitude of electrical charge, q, on the droplets is always an integral
multiple of the electrical charge, e, that is, q = n e, where n = 1, 2, 3, …..
Comments
Post a Comment